Search
- Page Path
- HOME > Search
Original Articles
- Thyroid
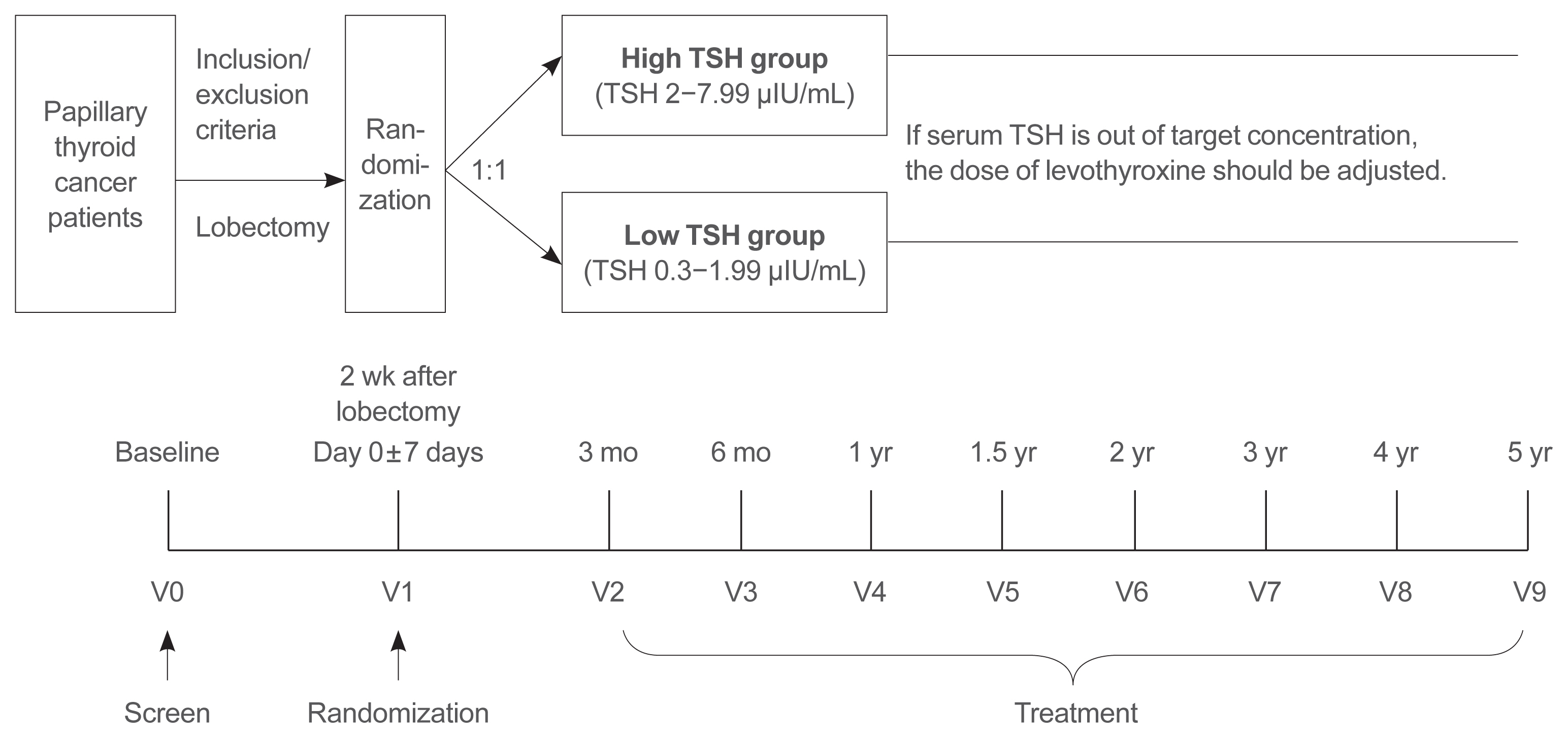
- A Multicenter, Randomized, Controlled Trial for Assessing the Usefulness of Suppressing Thyroid Stimulating Hormone Target Levels after Thyroid Lobectomy in Low to Intermediate Risk Thyroid Cancer Patients (MASTER): A Study Protocol
- Eun Kyung Lee, Yea Eun Kang, Young Joo Park, Bon Seok Koo, Ki-Wook Chung, Eu Jeong Ku, Ho-Ryun Won, Won Sang Yoo, Eonju Jeon, Se Hyun Paek, Yong Sang Lee, Dong Mee Lim, Yong Joon Suh, Ha Kyoung Park, Hyo-Jeong Kim, Bo Hyun Kim, Mijin Kim, Sun Wook Kim, Ka Hee Yi, Sue K. Park, Eun-Jae Jung, June Young Choi, Ja Seong Bae, Joon Hwa Hong, Kee-Hyun Nam, Young Ki Lee, Hyeong Won Yu, Sujeong Go, Young Mi Kang, MASTER study group
- Endocrinol Metab. 2021;36(3):574-581. Published online May 26, 2021
- DOI: https://doi.org/10.3803/EnM.2020.943
- 6,299 View
- 268 Download
- 8 Web of Science
- 11 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Background
Postoperative thyroid stimulating hormone (TSH) suppression therapy is recommended for patients with intermediate- and high-risk differentiated thyroid cancer to prevent the recurrence of thyroid cancer. With the recent increase in small thyroid cancer cases, the extent of resection during surgery has generally decreased. Therefore, questions have been raised about the efficacy and long-term side effects of TSH suppression therapy in patients who have undergone a lobectomy.
Methods
This is a multicenter, prospective, randomized, controlled clinical trial in which 2,986 patients with papillary thyroid cancer are randomized into a high-TSH group (intervention) and a low-TSH group (control) after having undergone a lobectomy. The principle of treatment includes a TSH-lowering regimen aimed at TSH levels between 0.3 and 1.99 μIU/mL in the low-TSH group. The high-TSH group targets TSH levels between 2.0 and 7.99 μIU/mL. The dose of levothyroxine will be adjusted at each visit to maintain the target TSH level. The primary outcome is recurrence-free survival, as assessed by neck ultrasound every 6 to 12 months. Secondary endpoints include disease-free survival, overall survival, success rate in reaching the TSH target range, the proportion of patients with major cardiovascular diseases or bone metabolic disease, the quality of life, and medical costs. The follow-up period is 5 years.
Conclusion
The results of this trial will contribute to establishing the optimal indication for TSH suppression therapy in low-risk papillary thyroid cancer patients by evaluating the benefit and harm of lowering TSH levels in terms of recurrence, metabolic complications, costs, and quality of life. -
Citations
Citations to this article as recorded by- Effect of thyroid-stimulating hormone suppression on quality of life in thyroid lobectomy patients: interim analysis of a multicenter, randomized controlled trial in low- to intermediate-risk thyroid cancer patients (MASTER study)
Ja Kyung Lee, Eu Jeong Ku, Su-jin Kim, Woochul Kim, Jae Won Cho, Kyong Yeun Jung, Hyeong Won Yu, Yea Eun Kang, Mijin Kim, Hee Kyung Kim, Junsun Ryu, June Young Choi
Annals of Surgical Treatment and Research.2024; 106(1): 19. CrossRef - Clinical impact of coexistent chronic lymphocytic thyroiditis on central lymph node metastasis in low- to intermediate-risk papillary thyroid carcinoma: The MASTER study
Da Beom Heo, Ho-Ryun Won, Kyung Tae, Yea Eun Kang, Eonju Jeon, Yong Bae Ji, Jae Won Chang, June Young Choi, Hyeong Won Yu, Eu Jeong Ku, Eun Kyung Lee, Mijin Kim, Jun-Ho Choe, Bon Seok Koo
Surgery.2024; 175(4): 1049. CrossRef - Dynamic Changes in Treatment Response af-ter 131I in Differentiated Thyroid Cancer and Their Relationship with Recurrence Risk Stratification and TNM Staging
璐 狄
Advances in Clinical Medicine.2024; 14(03): 1083. CrossRef - ASO Author Reflections: Active Surveillance may be Possible in Patients with T1b Papillary Thyroid Carcinoma Over 55 Years of Age Without High-Risk Features on Preoperative Examinations
Ho-Ryun Won, Eonju Jeon, Da Beom Heo, Jae Won Chang, Minho Shong, Je Ryong Kim, Hyemi Ko, Yea Eun Kang, Hyon-Seung Yi, Ju Hee Lee, Kyong Hye Joung, Ji Min Kim, Younju Lee, Sung-Woo Kim, Young Ju Jeong, Yong Bae Ji, Kyung Tae, Bon Seok Koo
Annals of Surgical Oncology.2023; 30(4): 2254. CrossRef - Outcomes and Trends of Treatments in High‐Risk Differentiated Thyroid Cancer
Arash Abiri, Khodayar Goshtasbi, Sina J. Torabi, Edward C. Kuan, William B. Armstrong, Tjoson Tjoa, Yarah M. Haidar
Otolaryngology–Head and Neck Surgery.2023; 168(4): 745. CrossRef - Current Controversies in Low-Risk Differentiated Thyroid Cancer: Reducing Overtreatment in an Era of Overdiagnosis
Timothy M Ullmann, Maria Papaleontiou, Julie Ann Sosa
The Journal of Clinical Endocrinology & Metabolism.2023; 108(2): 271. CrossRef - Age-Dependent Clinicopathological Characteristics of Patients with T1b Papillary Thyroid Carcinoma: Implications for the Possibility of Active Surveillance
Ho-Ryun Won, Eonju Jeon, Da Beom Heo, Jae Won Chang, Minho Shong, Je Ryong Kim, Hyemi Ko, Yea Eun Kang, Hyon-Seung Yi, Ju Hee Lee, Kyong Hye Joung, Ji Min Kim, Younju Lee, Sung-Woo Kim, Young Ju Jeong, Yong Bae Ji, Kyung Tae, Bon Seok Koo
Annals of Surgical Oncology.2023; 30(4): 2246. CrossRef - Potential impact of obesity on the aggressiveness of low- to intermediate-risk papillary thyroid carcinoma: results from a MASTER cohort study
Mijin Kim, Yae Eun Kang, Young Joo Park, Bon Seok Koo, Eu Jeong Ku, June Young Choi, Eun Kyung Lee, Bo Hyun Kim
Endocrine.2023; 82(1): 134. CrossRef - Differentiated thyroid cancer: a focus on post-operative thyroid hormone replacement and thyrotropin suppression therapy
Benjamin J. Gigliotti, Sina Jasim
Endocrine.2023; 83(2): 251. CrossRef - Thyroid stimulating hormone suppression and recurrence after thyroid lobectomy for papillary thyroid carcinoma
Mi Rye Bae, Sung Hoon Nam, Jong-Lyel Roh, Seung-Ho Choi, Soon Yuhl Nam, Sang Yoon Kim
Endocrine.2022; 75(2): 487. CrossRef - The Concept of Economic Evaluation and Its Application in Thyroid Cancer Research
Kyungsik Kim, Mijin Kim, Woojin Lim, Bo Hyun Kim, Sue K. Park
Endocrinology and Metabolism.2021; 36(4): 725. CrossRef
- Effect of thyroid-stimulating hormone suppression on quality of life in thyroid lobectomy patients: interim analysis of a multicenter, randomized controlled trial in low- to intermediate-risk thyroid cancer patients (MASTER study)

- Clinical Study
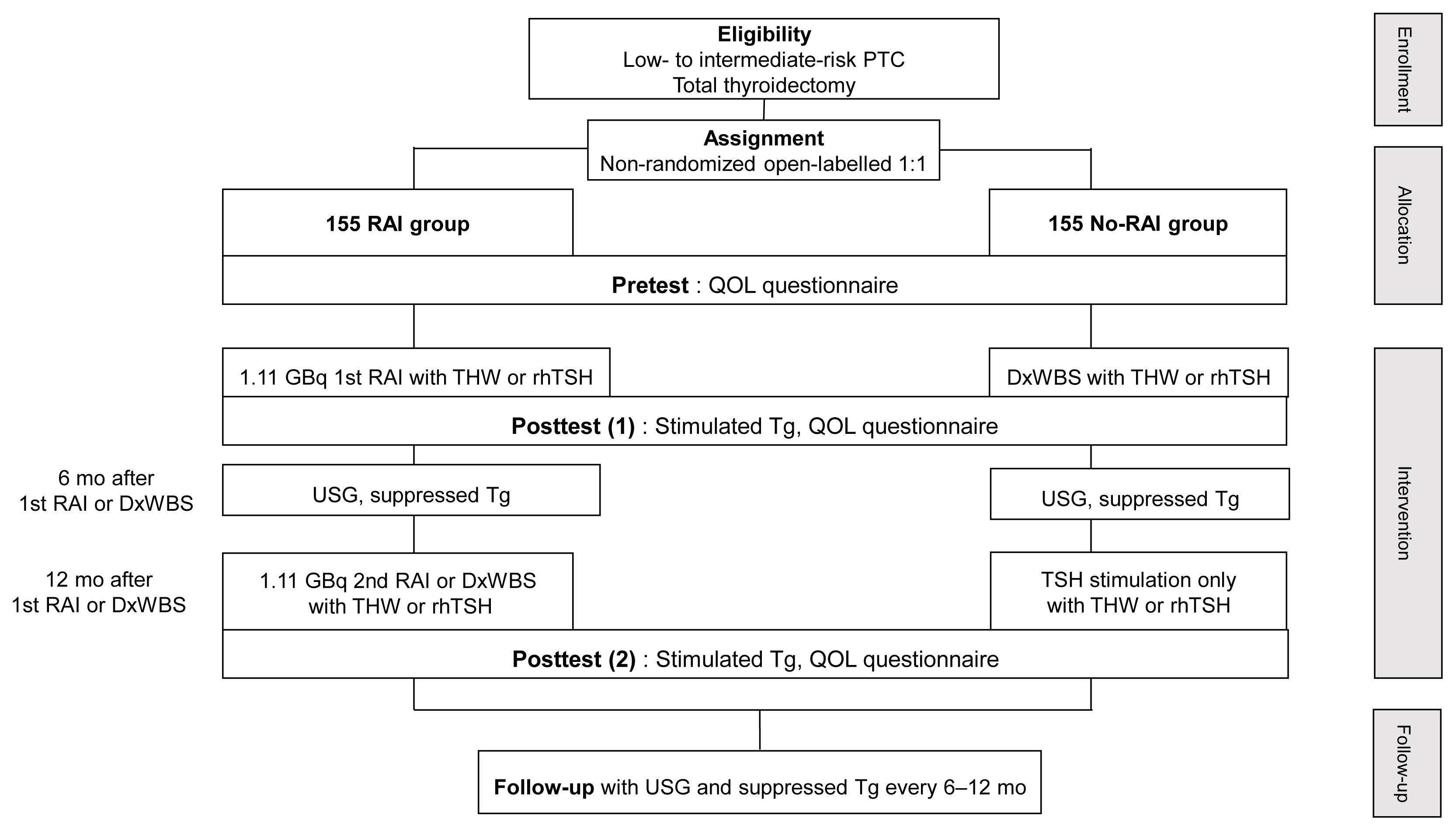
- A Phase II Multi-Center, Non-Randomized, Parallel Group, Non-Inferiority Study to Compare the Efficacy of No Radioactive Iodine Remnant Ablation to Remnant Ablation Treatment in Low- to Intermediate-Risk of Papillary Thyroid Cancer: The MOREthyroid Trial Protocol
- Eun Kyung Lee, You Jin Lee, Young Joo Park, Jae Hoon Moon, Ka Hee Yi, Koon Soon Kim, Joo Hee Lee, Sun Wook Cho, Jungnam Joo, Yul Hwangbo, Sujeong Go, Do Joon Park
- Endocrinol Metab. 2020;35(3):571-577. Published online September 22, 2020
- DOI: https://doi.org/10.3803/EnM.2020.681
- 4,640 View
- 119 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
Radioactive iodine (RAI) remnant ablation is recommended in patients with papillary thyroid cancer (PTC) and extrathyroidal extension or central lymph node metastasis. However, there exists little evidence about the necessity of remnant ablation in PTC patients with low- to intermediate-risk, those have been increasing in recent decades.
Methods
This multicenter, prospective, non-randomized, parallel group clinical trial will enroll 310 eligible patients with low- to intermediate-risk of thyroid cancer. Inclusion criteria are patients who recently underwent total thyroidectomy for PTC with 3 or less tumors of size 1≤ to ≤2 cm with no microscopic extension and N0/x, or size ≤2 cm with microscopic extension and/or N1a (number of lymph node ≤3, size of tumor foci ≤0.2 cm, and lymph node ratio <0.4). Patients choose to undergo RAI ablation (131I, dose 1.1 GBq) or diagnostic whole-body scan (DxWBS) (131I or 123I, dose 0.074 to 0.222 GBq), followed by subsequent measurement of stimulated thyroglobulin (sTg) within 1 year. Survey for quality of life (QOL) will be performed at baseline and at 1 year after follow-up. The total enrollment period is 5 years, and patients will be followed up for 1 year. The primary endpoint is the non-inferiority of surgery alone to surgery with ablation in terms of biochemical remission (BCR) rate (sTg ≤2 ng/mL) without evidence of structural recurrence. The secondary endpoint was the difference of QOL.
Conclusion
This study will evaluate whether surgery alone achieves similar BCR and improved QOL compared to RAI ablation in patients with low- to intermediate-risk PTC within 1 year.


 KES
KES

 First
First Prev
Prev



